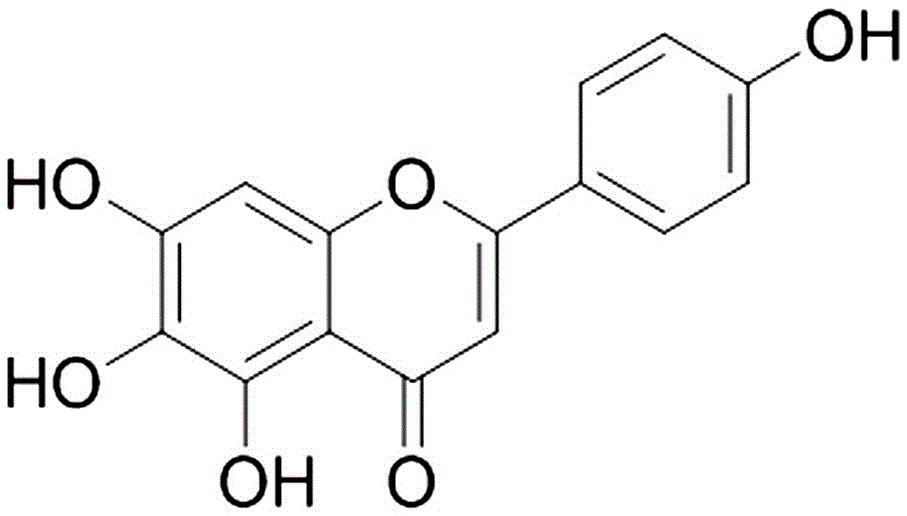
SCI论文(www.lunwensci.com):
摘要:无机砷是普遍存在的环境污染物,其通过饮用水引起人类多种疾病,我们介绍了无机砷通过激活Nrf2诱导的免疫性疾病的研究进展。
关键词:无机砷;Nrf2;免疫性疾病
本文引用格式:张金卫,陈万群.无机砷激活Nrf2诱发免疫性疾病研究进展[J].世界最新医学信息文摘,2019,19(66):108-110.
Advances in Research on Inorganic Arsenic-activated Nrf2-induced Immune Diseases
ZHANG Jin-wei,CHEN Wan-qun*
(Chongqing Hospital of Traditional Chinese Medicine,Chongqing)
ABSTRACT:Inorganic arsenic is a ubiquitous environmental pollutant.Inorganic arsenic causes many diseases through drinking water.We present the research progress of inorganic arsenic by activating Nrf2-induced immune diseases.
KEY WORDS:Inorganic arsenic;Nrf2;Immune diseases
0引言
无机砷是普遍存在的环境污染物,由无机砷污染的饮用水影响着全球超过1.4亿人[1],其中中国高达1466万[2],美国已经达到1300万[3]。研究表明,无机砷通过激活Nrf2通路诱导免疫系统疾病,下面详细介绍其研究进展。
1无机砷激活Nrf2的机制研究进展
1.1无机砷增强Keap1与Cul3相互作用
高剂量的无机砷引起急性毒性,而慢性无机砷暴露导致皮肤、肺脏、肾脏、肝脏、膀胱等器官中的肿瘤发生率升高,砷诱导DNA低甲基化并调节基因表达谱,这暗示了无机砷致癌作用的非基因毒性机制,因此研究由无机砷暴露引起的分子信号传导至关重要。异常表达的基因可以分为许多类别,如应激反应基因、激素相关基因、细胞因子、凋亡基因、细胞周期调控基因、蛋白水解基因和原癌基因[4-7]。在无机砷诱导的癌变过程中,控制细胞增殖和转化的基因发生异常表达[8-13]。
Keap1在多个水平上调节着Nrf2的活性。第一,Keap1能够感知细胞氧化还原条件中的干扰,从而相应地调节Nrf2信号传导途径。几种半胱氨酸残基在这种传感机制中起着重要作用,例如Keap1中C151的突变阻断了叔丁基对苯二酚或萝卜硫素诱导的Nrf2激活[14-16]。第二,Keap1作为E3泛素连接酶起作用,不断靶向Nrf2进行泛素化和降解,以使Nrf2的水平持续低表达[17-20]。第三,在诱导的水平上,E3泛素连接酶活性受到抑制,导致Nrf2降解减少和Nrf2核转位增强[18,21-22]。第四,在诱导后阶段,Keap1促进Nrf2核输出及其与细胞质泛素化和降解机制的关联以关闭Nrf2信号[23-26]。
Nrf2的泛素化受Keap1-Cul3 E3泛素连接酶复合物调节,其中Keap1作为底物适配子起作用。因此,含Keap1的E3泛素连接酶复合物的活性对于维持Nrf2的蛋白质水平至关重要。E3泛素连接酶复合物由Keap1、Cul3、Rbx1和E2组成。
亚砷酸盐、萝卜硫素和叔丁基对苯二酚都能激活Nrf2,在研究亚砷酸盐诱导Nrf2激活机制中发现:亚砷酸盐与萝卜硫素、叔丁基对苯二酚都是通过抑制Nrf2泛素化和降解来增强Nrf2蛋白水平,从而诱导Nrf2依赖性应答。然而,亚砷酸盐激活Nrf2作用机制却不同于萝卜硫素、叔丁基对苯二酚。亚砷酸盐明显的增强了Nrf2的E3泛素连接酶的亚基Keap1与Cul3之间的相互作用,这导致E3泛素连接酶的动态组装/解体受损,并因此降低其连接酶活性。
因此说明,亚砷酸盐激活Nrf2是不依赖于Keap1中的C151残基的,而萝卜硫素或叔丁基对苯二酚激活Nrf2是依赖于Keap1中的C151残基的[27]。
1.2Nrf2、Nrf1和Keap1共同参与无机砷诱发疾病的病理机制
首先利用慢病毒shRNA在人永生化角质细胞系HaCaT细胞中建立稳定敲低Nrf2(Nrf2-KD)和Keap1(Keap1-KD)角质细胞株。与正常组比较,Nrf2-KD细胞组的Nrf2及其调控的HMOX-1、NQO1、GCLC、GCLM基因表达显著下调;与正常组相比,Keap1-KD细胞组ARE依赖性基因表达上调,例如HMOX-1、NQO1、SRX1、GCLC和GCLM。
亚砷酸盐分别诱导正常角质细胞、Nrf2-KD角质细胞和Keap1-KD角质细胞发现:然而Keap1的缺乏减少了亚砷酸盐诱导的HMOX-1的mRNA和蛋白水平的表达水平。此外,在Keap1-KD组中未观察到亚砷酸盐诱导的其他ARE依赖性基因表达的增加。提示Keap1的稳定敲低导致ARE依赖性基因的基础表达增强,但弱化亚砷酸盐诱导的抗氧化反应。同时表明,组成性激活Nrf2可能会减弱ARE依赖性基因表达,特别是HMOX-1,以应对急性亚砷酸盐的暴露。
缺乏Nrf2导致Keap1的基因和蛋白表达减弱,这说明了Keap1的表达依赖于Nrf2。Nrf2的缺乏没有增加Nrf1的mRNA表达,但Nrf2敲低细胞在高浓度的亚砷酸盐暴露下表现出Nrf1的蛋白质积累增加(2.5-20M,6小时;10M,6-24小时),提示Nrf1可以补偿亚砷酸盐暴露条件下Nrf2的缺乏。此外,在正常细胞中观察到Nrf1蛋白水平的上调,但与Nrf2敲低细胞中观察到的持续反应相比是短暂的。相比之下,Keap1的敲低增强了Nrf2在正常和亚砷酸盐暴露条件下的蛋白质积累。Keap1敲低细胞表现出由高浓度亚砷酸盐诱导的Nrf1蛋白水平降低,但mRNA表达无变化。
在Keap1-KD细胞中,高浓度的亚砷酸盐诱导Nrf1蛋白表达微弱的升高,同时伴有Nrf2蛋白的显著升高,并且Nrf1的mRNA和蛋白质表达不一致。因此说明,由高浓度亚砷酸盐诱导Keap1-KD细胞Nrf1蛋白的较弱水平的积累可能是由于Nrf2介导的。另外,Nrf1的mRNA和蛋白质表达之间的差异表明蛋白质稳定是Nrf1活化的主要机制[28]。
亚砷酸盐分别诱导Nrf2-KD细胞和Keap1-KD细胞24小时,发现Nrf2敲低细胞明显提高了亚砷酸盐毒性的敏感性和凋亡率,而Keap1敲低细胞对亚砷酸盐毒性以及凋亡表现为显著的抗性。为了进一步证明,Nrf2激活促进了亚砷酸盐诱导凋亡,用Nrf2激活剂叔丁基氢醌预先干预亚砷酸盐诱导的Nrf2敲低的细胞,结果发现叔丁基氢醌对亚砷酸盐的毒性没有抑制作用,表明叔丁基氢醌的保护作用源于Nrf2的激活。而且,在Keap1敲低的细胞中,叔丁基氢醌也没有体现保护作用。相反,用非细胞毒性剂量的亚砷酸盐(2.5M)预处理,却体现出稍微保护作用。虽然低浓度的亚砷酸盐预处理正常细胞可以防止随后由更高剂量的亚砷酸盐诱导的细胞毒性,但是在Nrf2敲低细胞中也存在保护作用,这表明低剂量亚砷酸盐可能诱发了除Nrf2之外的其他适应性免疫机制,例如激活Nrf1。与叔丁基氢醌预处理类似,低浓度亚砷酸盐预处理并没有保护Keap1敲低细胞免受高浓度亚砷酸盐的毒性。
因此表明,Nrf2、Keap1和Nrf1相互作用并协调参与亚砷酸盐诱导的皮肤毒性反应[29]。
2无机砷激活Nrf2诱发免疫相关性疾病的研究进展
砷是一种免疫抑制金属,其损害许多关键器官。研究表明低浓度的无机砷不仅抑制人体外周血单核细胞向功能性巨噬细胞分化,还逆转人巨噬细胞的主要表型和遗传特征[30,31]。研究表明,无机砷在多种器官或者细胞中激活Nrf2,例如肝脏、肾脏、角质细胞、支气管上皮细胞等[32-35]。下面从体内研究和体外研究介绍无机砷激活Nrf2诱发免疫相关疾病的研究进展。
2.1无机砷激活Nrf2诱发免疫相关疾病的体内研究
亚砷酸盐刺激C57BL/6小鼠6小时,引起小鼠脾脏、胸腺以及外周血单核细胞的Nrf2高表达,并上调Nrf2的下游基因HO-1、胱甘肽S转移酶(glutathione-S-transferase,GST)、氨酸-半胱氨酸连接酶(glutamate-cysteine ligase,GCL)和谷胱甘肽还原酶(glutathione reductase,GR)的蛋白表达。另外,脾脏和胸腺的NQO1、谷氨酸-半胱氨酸连接酶催化酶(Glutamate-cysteine ligase catalytic,Gclc)和GCL的mRNA表达上调。因此表明,急性亚砷酸盐暴露诱发了免疫系统的Nrf2激活[36]。
2.2无机砷激活Nrf2诱发免疫相关疾病的体外研究
2.2.1在无机砷诱发的淋巴性疾病中,Nrf2是介导砷毒性早期信号通路
基因芯片微阵列分析是研究外部刺激、内部刺激诱导的全基因表达异常的重要工具,常用来鉴定细胞应答的信号传导途径。5M亚砷酸盐诱导CL-1人非肿瘤淋巴母细胞(CL-1 human non-tumoral lymphoblastoid cells)0、2和12小时,用基因芯片法检测基因表达发现:与正常组比较,亚砷酸盐诱导组共有299个基因表达异常,其中在亚砷酸盐诱导2小时组中81个基因发生了改变(33个基因下调和48个基因上调),在亚砷酸盐诱导12小时组中218个基因发生了改变(153个基因下调和65个基因上调);有48个基因在亚砷酸盐诱导2小时组改变最大,有185个基因在亚砷酸盐诱导12小时组改变最大,有33个基因在亚砷酸盐诱导2小时和12小时组改变都非常大。用qRT-PCR验证上述基因芯片检测结果发现:qRT-PCR检测结果与基因芯片检测结果一致。而且,用qRT-PCR法在淋巴母细胞系CL-45细胞(lymphoblastoid cell line,CL-45)和原代的淋巴母细胞(primary lymphoblastoid cells)重现了上述结果。
在分析调节这些基因改变的信号通路时发现,亚砷酸盐诱导2小时和12小时组,Nrf2调控的靶基因改变都是最明显的;亚砷酸盐诱导2小时组涉及的其他信号通路还包括“干扰素信号传导”、“B细胞受体信号传导”、“糖皮质激素受体信号传导”和“FCγ受体信号传导途径”;亚砷酸盐诱导12小时组涉及的其他信号通路包括“芳基烃受体信号”、“谷胱甘肽代谢”和“谷氨酸代谢”等。用免疫印迹法从蛋白水平上检测Nrf2通路及其下游蛋白表达的情况发现:亚砷酸盐诱导Nrf2蛋白及其调控的HMOX1蛋白、NQO1蛋白表达升高。另外,在qRT-PCR实验中发现:Nrf2的靶基因HMOX1是亚砷酸盐诱导上调最高的基因,其mRNA的表达在亚砷酸盐诱导12小时组0.1-5mM浓度梯度中呈现剂量依赖性上调,而且其表达在5mM亚砷酸盐诱导组20-120分钟时间梯度中呈现时间依赖性上调。此外,其基因的上调没有随着亚砷酸盐毒性的累计而减弱。
另外,研究表明,亚砷酸盐激活Nrf2调控的基因改变决定于诱导的细胞类型,例如在单核细胞、角质形成细胞、肾脏、内皮细胞和尿路上皮细胞中HMOX1、NQO1、GCLM和GCLC基因上调[6,37-40];在单核细胞和角质形成细胞中FTL和FTH1基因表达上调[39-41]。
因此表明,在亚砷酸诱导淋巴性疾病中,Nrf2是介导砷毒性早期信号通路中最重要的途径;另外,“干扰素信号传导”、“B细胞受体信号传导”、“糖皮质激素受体信号传导”、“FCγ受体信号传导”、“芳基烃受体信号传导”、“谷胱甘肽代谢”和“谷氨酸代谢”也有可能参与砷毒性的信号传导[42]。
2.2.2在无机砷诱导的小鼠胚胎成纤维细胞中,Nrf2是介导砷毒性的早期信号通路
亚砷酸盐诱导正常的小鼠胚胎成纤维细胞(murine embryonic fibroblasts,NEF)发现:Nrf2表达呈时间和剂量依赖性上调。亚砷酸盐分别诱导正常的NEF细胞和Nrf2缺陷型的NEF细胞发现:两组HO-1蛋白(HO-1是Nrf2的靶基因)表达都升高,但是,正常组比Nrf2缺陷组高出2倍;而且,正常组HO-1蛋白在亚砷酸盐诱导2时开始上调,12小时达到最高峰,24小时明显降低,而Nrf2缺陷组HO-1蛋白在亚砷酸盐诱导4小时开始上调,12小时最高峰,24小时仍然没有下调;正常组HO-1的mRNA表达在亚砷酸盐诱导1小时开始上调,6小时达到最高峰,随后开始降低,而Nrf2缺陷组HO-1的mRNA表达在亚砷酸盐诱导1小时开始上调,6小时最高峰,24小时依然不减。因此说明,激活Nrf2在亚砷酸盐诱导的小鼠胚胎成纤维细胞的早期起着重要的作用[43]。
2.2.3无机砷激活Nrf2调控S100A9诱发免疫相关疾病
无机砷破坏各种类型的免疫细胞的功能,导致器官的功能紊乱,甚至发生癌变。以往的研究表明,S100A9诱导了自身免疫疾病、心血管疾病以及癌症[44,45]。在研究亚砷酸盐通过激活Nrf2调控S100A9的实验中,用亚砷酸盐刺激肺上皮A549细胞(lung epithelial A549 cells)、肾癌Caki-2细胞(kidney carcinoma Caki-2 cells)、胚胎肾HEK293细胞(embryonic kidney HEK293 cells)、肝癌HepG2细胞(liver carcinoma HepG2 cells)、永生化T淋巴Jurkat细胞(immortalized T lymphocyte Jurkat cells)、人永生化皮肤角质形成细胞系HaCaT细胞(immortalized skin keratinocytic HaCaT cells)、正常人表皮角质形成细胞(normal human epidermal keratinocytes,NHEK)、白血病单核细胞淋巴瘤U937细胞(leukemic monocyte lymphoma U937 cells)和永生化膀胱尿路上皮UROtsa细胞(immortalized bladder urothelial UROtsa cells)1-2周发现:在HaCaT细胞、UROtsa细胞和U937细胞中S100A9的mRNA表达显著上调,同时伴有Nrf2的高表达,但在其他细胞中没有变化。为了研究亚砷酸盐上调S100A9的机制,将Nrf2不同的片段转入HaCaT细胞,荧光霉素报告基因实验发现:激活的Nrf2与S100A9相互作用,并上调了S100A9的表达。因此表明,亚砷酸盐通过激活Nrf2调控S100A9的表达,从而诱发免疫相关疾病[46]。
参考文献
[1]L.Rodriguez-Lado,G.Sun,M.Berg,et al.Johnson,Groundwater arsenic contamination throughout China[J].Science(New York,N.Y.),2013,(341):866-868.
[2]G.Sun,X.Li,J.Pi,Y.Sun,B.Li,Y.Jin,Y.Xu.Current research problems of chronic arsenicosis in China[J].Journal of health,population,and nutrition,2006,(24):176-181.
[3]oods,E.Yehuda-Shnaidman,Q.Zhang,V.et al.Low-level arsenic impairs glucose-stimulated insulin secretion in pancreatic beta cells:involvement of cellular adaptive response to oxidative stress[J].Environmental health perspectives,2010,(118):864-870.
[4]L.H.Yih,K.Peck,T.C.Lee.Changes in gene expression profiles of human fibroblasts in response to sodium arsenite treatment[J].Carcinogenesis,2002,(23):867-876.
[5]H.Chen,S.Li,J.Liu,B.A.Diwan,J.C.et al.Waalkes,Chronic inorganic arsenic exposure induces hepatic global and individual gene hypomethylation:implications for arsenic hepatocarcinogenesis[J].Carcinogenes is,2004,(25):1779-1786.
[6]X.H.Zheng,G.S.Watts,S.Vaught,A.J.Gandolfi,Low-level arsenite induced gene expression in HEK293 cells[J].Toxicology,2003,(187):39-48.
[7]K.T.Kitchin,Recent advances in arsenic carcinogenesis:modes of action,animal model systems,and methylated arsenic metabolites[J].Toxicology and applied pharmacology,2001,172:249-261.
[8]H.Chen,J.Liu,C.Q.et al.Waalkes,Association of c-myc overexpression and hyperproliferation with arsenite-induced malignant transformation[J].Toxicology and applied pharmacology,2001,175:260-268.
[9]H.K.Hamadeh,K.J.Trouba,R.P.Amin,C.et al.Coordination of altered DNA repair and damage pathways in arsenite-exposed keratinocytes[J].Toxicological sciences:an official journal of the Society of Toxicology,2002,(69):306-316.
[10]M.Shimizu,J.F.Hochadel,B.A.Fulmer,M.P.Waalkes,Effect of glutathione depletion and met allothionein gene expression on arsenic-induced cytotoxicity and c-myc expression in vitro[J].Toxicological sciences
:an official journal of the Society of Toxicology,1998,(45):204-211.
[11]J.Liu,Y.Xie,J.M.Ward,B.A.Diwan,M.P.Waalkes,Toxicogenomic analysis of aberrant gene expression in liver tumors and nontumorous livers of adult mice exposed in utero to inorganic arsenic[J].Toxicological sciences:an official journal of the Society of Toxicology,2004,(77):249-257.
[12]J.Li,M.Gorospe,J.Barnes,Y.Liu.Tumor promoter arsenite stimulates histone H3 phosphoacetylation of proto-oncogenes c-fos and c-jun chromatin in human diploid fibroblasts[J].The Journal of biological chemistry,2003,(278):13183-13191.
[13]L.Benbrahim-Tallaa,R.A.Waterland,M.Styblo,W.E.et al.Molecular events associated with arsenic-induced malignant transformation of human prostatic epithelial cells:aberrant genomic DNA methylation and K-ras oncogene activation[J].Toxicology and applied pharmacology,
[14]N.Wakabayashi,A.T.Dinkova-Kostova,W.D.Holtzclaw,M.I.Kang,A.et al.Protection against electrophile and oxidant stress by induction of the phase 2 response:fate of cysteines of the Keap1 sensor modified by inducers[J].Proceedings of the National Academy of Sciences of the United States of America,2004,(101):2040-2045.
[15]A.L.Levonen,A.Landar,A.Ramachandran,E.K.Ceaser,D.A.et al.Darley-Usmar,Cellular mechanisms of redox cell signalling:role of cysteine modification in controlling antioxidant defences in response to electrophilic lipid oxidation products[J].The Biochemical journal,2004,(378):373-382.
[16]D.D.Zhang,M.Hannink.Distinct cysteine residues in Keap1 are required for Keap1-dependent ubiquitination of Nrf2 and for stabilization of Nrf2 by chemopreventive agents and oxidative stress[J].Mol Cell Biol
,2003,(23):8137-8151.
[17]M.Furukawa,Y.Xiong.BTB protein Keap1 targets antioxidant transcription factor Nrf2 for ubiquitination by the Cullin 3-Roc1 ligase[J].Mol Cell Biol,2005,(25):162-171.
[18]D.D.Zhang,S.C.Lo,J.V.Cross,D.J.Templeton,M.Hannink,Keap1 is a redox-regulated substrate adaptor protein for a Cul3-dependent ubiquitin ligase complex[J].Mol Cell Biol,2004,(24):10941-10953.
[19]A.Kobayashi,M.I.Kang,H.Okawa,M.et al.Oxidative stress sensor Keap1 functions as an adaptor for Cul3-based E3 ligase to regulate proteasomal degradation of Nrf2[J].Mol Cell Biol,2004,(24):7130-7139.
[20]S.B.Cullinan,J.D.Gordan,J.Jin,J.W.Harper,J.A.Diehl,The Keap1-BTB protein is an adaptor that bridges Nrf2 to a Cul3-based E3 ligase:oxidative stress sensing by a Cul3-Keap1 ligase[J].Mol Cell Biol,2004,(24):8477-8486.
[21]D.D.Zhang,Mechanistic studies of the Nrf2-Keap1 signaling pathway[J].Drug metabolism reviews,2006,(38):769-789.
[22]A.K.Jain,D.A.Bloom,A.K.Jaiswal,Nuclear import and export signals in control of Nrf2[J].The Journal of biological chemistry,2005,(280):29158-29168.
[23]T.Nguyen,P.J.Sherratt,P.Nioi,et al.Nrf2 controls constitutive and inducible expression of ARE-driven genes through a dynamic pathway involving nucleocytoplasmic shuttling by Keap1[J].The Journal of biological chemistry,2005,(280):32485-32492.
[24]Z.Sun,S.Zhang,J.Y.Chan,D.D.Zhang,Keap1 controls postinduction repression of the Nrf2-mediated antioxidant response by escorting nuclear export of Nrf2[J].Mol Cell Biol,2007,(27):6334-6349.
[25]R.N.Karapetian,A.G.Evstafieva,I.S.Abaeva,N.V.Chichkova,G.S.Filonov,Y.P.Rubtsov,E.A.Sukhacheva,S.V.Melnikov,U.Schneider,E.
E.Wanker,A.B.Vartapetian,Nuclear oncoprotein prothymosin alpha is a partner of Keap1:implications for expression of oxidative stress-protecting genes[J].Mol Cell Biol,2005(25):1089-1099.
[26]M.Velichkova,T.Hasson,Keap1 regulates the oxidation-sensitive shuttling of Nrf2 into and out of the nucleus via a Crm1-dependent nuclear export mechanism[J].Mol Cell Biol,2005,(25):4501-4513.
[27]X.J.Wang,Z.Sun,W.Chen,Y.Li,N.F.Villeneuve,D.D.Zhang,Activation of Nrf2 by arsenite and monomethylarsonous acid is independent of Keap1-C151:enhanced Keap1-Cul3 interaction[J].Toxicology and applied pharmacology,2008,(230):383-389.
[28]J.Steffen,M.Seeger,A.Koch,E.Kruger,Proteasomal degradation is transcriptionally controlled by TCF11 via an ERAD-dependent feedback loop[J].Molecular cell,2010(40):147-158.
[29]R.Zhao,Y.Hou,Q.Zhang,C.G.Woods,P.et al.Cross-regulations among NRFs and KEAP1 and effects of their silencing on arsenic-induced antioxidant response and cytotoxicity in human keratinocytes[J].Environmental health perspectives,2012,(120):583-589.
[30]A.Lemarie,C.Morzadec,D.Merino,O.et al.Arsenic trioxide induces apoptosis of human monocytes during macrophagic differentiation through nuclear factor-kappaB-related survival pathway down-regulation[J].The Journal of pharmacology and experimental therapeutics,2006,(316):304-314.
[31]A.Lemarie,C.Morzadec,E.Bourdonnay,O.et al.Human macrophages constitute targets for immunotoxic inorganic arsenic[J].Journal of immunology,2006,(177):3019-3027.
[32]S.Sarkar,S.Mukherjee,A.Chattopadhyay,S.Bhattacharya,Low dose of arsenic trioxide triggers oxidative stress in zebrafish brain:expression of antioxidant genes[J].Ecotoxicology and environmental safety,2014,(107):1-8.
[33]S.M.Prabu,M.Muthumani,Silibinin ameliorates arsenic induced nephrotoxicity by abrogation of oxidative stress,inflammation and apoptosis in rats[J].Molecular biology reports,2012,(39):11201-11216.
[34]J.Pi,W.Qu,J.M.Reece,Y.et al.Transcription factor Nrf2 activation by inorganic arsenic in cultured keratinocytes:involvement of hydrogen peroxide[J].Exp Cell Res,2003,(290):234-245.
[35]G.Wang,P.Xiu,F.Li,C.Xin,K.Li,Vitamin A supplementation alleviates extrahepatic cholestasis liver injury through Nrf2 activation[J].Oxidative medicine and cellular longevity,2014,273692.
[36]X.Duan,J.Li,Y.Zhang,W.et al.Activation of NRF2 pathway in spleen,thymus as well as peripheral blood mononuclear cells by acute arsenic exposure in mice[J].International immunopharmacolo gy,2015,(28):1059-1067.
[37]K.A.Bailey,S.D.Hester,G.W.Knapp,et al.Thai,Gene expression of normal human epidermal keratinocytes modulated by trivalent arsenicals[J].Molecular carcinogenesis,2010,(49):981-998.
[38]Y.Fujiwara,A.Honda,M.Satoh,DNA microarray gene expression analysis of human vascular endothelial cells exposed to arsenite[J].The Journal of toxicological sciences,2010,(35):275-278.
[39]P.F.Su,Y.J.Hu,I.C.Ho,et al.Distinct gene expression profiles in immortalized human urothelial cells exposed to inorganic arsenite and its methylated trivalent metabolites[J].Environmental health perspectiv es,2006(114):394-403.
[40]C.Martin-Chouly,C.Morzadec,M.Bonvalet,M.D.Galibert,O.Fardel,
L.Vernhet,Inorganic arsenic alters expression of immune and stress response genes in activated primary human T lymphocytes[J].Molecular immunology,2011,(48):956-965.
[41]M.A.Rea,J.P.Gregg,Q.Qin,et al.Global alteration of gene expression in human keratinocytes by inorganic arsenic[J].Carcinogenes is,2003(24):747-756.
[42]E.J.Cordova,A.Martinez-Hernandez,L.Uribe-Figueroa,F.et al.The NRF2-KEAP1 pathway is an early responsive gene network in arsenic exposed lymphoblastoid cells[J].PloS one,2014,9:e88069.
[43]H.Harada,R.Sugimoto,A.Watanabe,S.et al.Differential roles for Nrf2 and AP-1 in upregulation of HO-1 expression by arsenite in murine embryonic fibroblasts[J].Free radical research,2008,(42):297-304.
[44]W.Nacken,J.Roth,C.Sorg,C.Kerkhoff,S100A9/S100A8:Myeloid representatives of the S100 protein family as prominent players in innate immunity[J].Microscopy research and technique,2003,(60):569-580.
[45]J.M.Ehrchen,C.Sunderkotter,D.Foell,T.Vogl,J.Roth,The endogenous Toll-like receptor 4 agonist S100A8/S100A9(calprotectin)as innate amplifier of infection,autoimmunity,and cancer[J].Journal of leukocyte biology,2009,(86):557-566.
[46]D.Sumi,Y.Shimizu,S.Himeno,Involvement of Nrf2 activation in the upregulation of S100A9 by exposure to inorganic arsenite[J].International journal of molecular medicine,2013,(31):259-264.
关注SCI论文创作发表,寻求SCI论文修改润色、SCI论文代发表等服务支撑,请锁定SCI论文网! 文章出自SCI论文网转载请注明出处:https://www.lunwensci.com/yixuelunwen/19079.html