SCI论文(www.lunwensci.com):
摘要:cGMP依赖性蛋白激酶I(cGMP-dependent protein kinases I,PKGI)参与结肠癌、乳腺癌、卵巢癌等多种肿瘤的发生、发展过程。然而,因PKGI存在两种亚型,即PKGIα和PKGIβ,且其在不同肿瘤组织中呈现抗肿瘤或促肿瘤的不同效应,为深入研究PKGI在肿瘤发病机制中的作用构成了极大困扰。本文依托cGMP/PKGI信号通路,综述PKGI在肿瘤研究中的最新进展,为进一步阐释PKGI(PKGIα和PKGIβ)在肿瘤发病机制中的作用提供参考,同时为靶向cGMP/PKGI信号通路的抗肿瘤药物研发提供理论依据。
关键词:肿瘤;cGMP依赖的蛋白激酶I(PKGI);抗肿瘤
本文引用格式:张毅峰,陆肖枫.cGMP依赖性蛋白激酶I在肿瘤中的研究进展[J].世界最新医学信息文摘,2019,19(98):125-127,131.
Adances in Study of cGMP-dependent Protein Kinases I in Cancers
ZHANG Yi-feng,LU Xiao-feng*
(Suzhou Ninth People's Hospital,Suzhou Jiangsu)
ABSTRACT:cGMP-dependent protein kinase I(PKGI)is involved in the development and progression of colon cancer,breast can-cer,ovarian cancer and other cancers.However,there are two subtypes of PKGI(PKGIαand PKGIβ),exhibiting different biological effects of anti-cancer or cancer-promoting in different tumors,which causes great difficulty in finding the deep mechanisms of PKGI(PKGIαand PKGIβ)in the pathogenesis of cancers.This article relies on cGMP/PKGI signal pathway,reviewing the latest advances about PKGI in cancer research,which can furnish a reference for further explanation of the role of PKGI in cancer pathogenesis and provide a theoretical basis for development of anti-cancer drugs targeted cGMP/PKGI signal pathway.
KEY WORDS:Cancer;PKGI;Anticancer
0引言
环鸟苷-3,5-单磷酸(cGMP)依赖性蛋白激酶又称蛋白激酶G(cGMP-dependent protein kinases,PKG),属于丝氨酸/苏氨酸激酶家族,广泛存在于真核细胞中[1]。PKG与底物cGMP结合后被激活,可引起一系列生理变化,如舒张血管平滑肌、抑制血小板聚集、细胞骨架重构等[2,3]。研究表明,PKGI与多种肿瘤的发生、发展密切相关,如乳腺癌、结肠癌、卵巢癌、胰腺癌、肺癌等[4-8]。体内外研究显示,PKGIα和PKGIβ在不同的肿瘤细胞中发挥不同的生物学效应,如激活或过表达PKGIβ能够抑制结肠癌细胞的增殖[9],而激活卵巢癌和胰腺癌细胞中PKGIα的活性则可抑制细胞凋亡,促进细胞增殖[7,10]。这提示PKGI具有抗肿瘤和促肿瘤的双重作用,但具体机制尚不明确。
1PKG I
1.1PKG I结构
在哺乳动物体内,PKGI主要分布在血管平滑肌、肠道平滑肌、血小板、内皮细胞、纤维母细胞等组织细胞中,而PKGII则多局限在脑和肾脏中[2,11]。PKGI由10号染色体上的I型基因编码并通过基因的交错拼接生成两种亚型(PKGIα和PKGIβ),PKGII则由坐落在4号染色体上的II型基因编码[12,13]。PKGIα和PKG Iβ在结构上高度同源,仅氨基末端的84和104位氨基酸残基不同[12]。PKGI由三个结构域组成:N末端,调节结构域,催化结构域。调节结构域包含两个串联的cGMP结合位点,当两个结合位点均结合cGMP时,PKGI被激活并启动下游信号级联放大反应,引起一系列的生物学效应[14,15]。
1.2PKG I的激活及生理功能
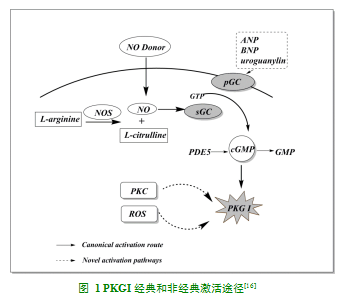
细胞内NO-sGC-cGMP-PKGI信号通路是PKGI激活的经典途径,同时细胞外的一些肽类激素,如心房肽、脑钠肽和尿鸟苷素等也可激活细胞膜上的微粒鸟苷酸环化酶(pGC),以另一种经典途径激活PKGI[16]。此外,PKGI亦可被活性氧(reactive oxygen species,ROS)和蛋白激酶C(protein kinase C)直接磷酸化以非经典途径激活[16,17]。活化后的PKGI通过激活BKca离子通道[20]、肌球蛋白轻链磷酸酶、磷酸化1,4,5-三磷酸肌醇受体,下调细胞内的Ca2+水平,舒张血管平滑肌,抑制血小板聚集[18,19];也可直接调控FHOD1(FHdomain-containing protein 1)[20]、波形蛋白(vimentin)[21,22]和血管扩张刺激磷蛋白(vasodilator stimulated phosphoprotein,VASP)[23],参与细胞骨架重构和细胞迁移。磷酸二酯酶5(phosphodiesterase 5,PDF5)能够特异性识别 cGMP,水解 cGMP 生成 GMP,抑制 PKGI 的激活[2(4]见图 1)。
2PKGI 与肿瘤的关系
2.1PKGI 与结肠癌的关系
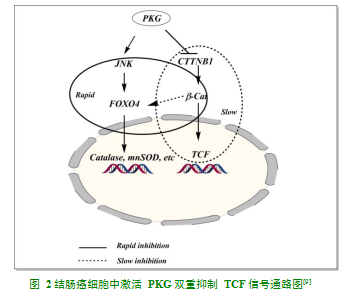
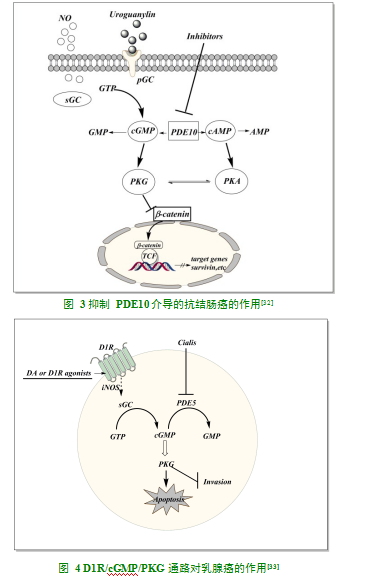
体外研究发现,在SW480和LoVo结肠癌细胞系中过表达PKGIβ,可以在启动子活性和蛋白表达水平上增强抑癌基因Rb、p27KIPI、p21CIPI的转录活性和蛋白表达量,诱导细胞凋亡[25]。此外,体内研究显示,过表达PKGI可下调SW620结肠癌细胞[26]移植瘤裸鼠肿瘤中的β-catenin,抑制肿瘤血管生成。同时,研究发现,抑制β-catenin可增加肿瘤细胞的失巢敏感性,而高表达PKGI的SW480细胞,其失巢率增加4倍[27],这提示过表达PKGI也可以通过下调β-catenin,增强结肠癌细胞的失巢敏感性。另有研究发现,给予WiDr结肠癌细胞低剂量的外源性NO,可通过激活NO-cGMP-PKG-ERK1/2-AP1-FRA1/2-MMP2/9信号通路,促进肿瘤的迁移侵袭[28]。此通路虽未涉及到PKGI,但文献报道激活PKGIα具有抗凋亡和细胞保护作用[29],同时,PKGIα对cGMP的敏感性是PKGIβ的10倍[16],因此,外源性低剂量NO可能通过选择性激活细胞内PKGIα介导WiDr结肠癌细胞的迁移侵袭,具体机制有待进一步研究。近期研究发现,在SW480和HCT116结肠癌细胞中过表达PKG能够双重抑制TCF(T-cell factor),这种抑制作用分为慢抑制和快速抑制,在慢抑制过程中,PKG通过抑制CTTNB1基因,下调β-catenin表达,抑制TCF的激活;而在快速抑制过程中,PKG通过激活JNK(cJun-N-terminal kinase),进而活化FOXO(forhead box O),活化的FOXO一方面快速募集β-catenin,另一方面入核启动基因表达p27、GADD45、过氧化氢酶和mnSOD等,最终抑制结肠癌细胞增殖,促进细胞凋亡[9,30](见图2)。此外,PKGI能够快速抑制TCF的激活[16],因此上述通路可能和PKGI密切关系,但具体作用机制尚不十分明确。磷酸二酯酶10A(phosphodiesterase 10A,PDE10)是PDE亚型中的一种,可非特异性水解cGMP和cAMP,临床多用于治疗精神分裂症和亨廷顿舞蹈病。体内研究显示,在人结肠癌样本和Apc(Min/+)小鼠结肠癌组织中PDE10过度表达,siRNA基因沉默PDE10或使用PDE10小分子抑制剂,均可选择性抑制结肠癌细胞的生长,而对正常结肠细胞无影响[31],体外进一步研究发现,在HT-29、HCT116和SW480中敲除PDE10,可上调cGMP和cAMP,激活PKG和PKA,抑制β-catenin入核,进而下调β-catenin介导的转录(如survivin等),诱导细胞凋亡(见图3)[32]。这一过程可能与过度激活结肠癌细胞中PKGIβ相关,具体机制有待进一步研究。综上,激活PKG对结肠癌细胞具有双重效应,即抗肿瘤和促肿瘤作用,这种效应可能由以下原因引起:1.激活方式不同,低剂量NO激活能力弱,主要激活对cGMP敏感的PKGIα,因此具有促肿瘤的效应,而过表达PKG或者抑制PDE10,可以很大程度激活PKG Iβ,而表现抗肿瘤效应;2.不同结肠癌细胞系PKGI及其亚型表达水平不同。
2.2PKGI与乳腺癌的关系
体外研究发现,在MCF-7和MDA-MB-468乳腺癌细胞中,使用8-Br-PET-cGMP选择性激活PKGIβ,能够抑制乳腺癌细胞生长,促进细胞凋亡,而选择性激活PKGIα(8-APT-cGMP)和PKGII(8-pCPT-cGMP)对细胞凋亡无影响[5]。研究数据显示,多巴胺-1型受体(DA type-1 receptor,D1R)在乳腺肿瘤组织和乳腺癌细胞系中均有表达,多巴胺(DA)或D1R受体激动剂可以通过激活D1R/cGMP/PKG信号通路,抑制乳腺细胞侵袭能力和细胞活力,诱导细胞凋亡,而在激活D1R受体的同时,给予PDE5抑制剂,能够显著增强这一抗肿瘤效应(图4)[33]。这种效应可能与cGMP升高水平有关,同时给予D1R受体激动剂和PDE5抑制剂可以在很大程度上提高cGMP的量,激活对cGMP不敏感的PKGIβ,发挥抗肿瘤的作用。另有研究发现,在MDA-MB-231和293T乳腺癌细胞中,激活PKGIβ可通过磷酸化钙调结合蛋白(caldesmon,CaD),破坏Caldesmon–Myosin之间相互作用,增加乳腺癌细胞的侵袭和迁移能力[34]。这种差异可能是由于细胞种类、培养条件、细胞代数不同等引起的,总的来讲,激活PKG(尤其是PKGIβ)具有抗肿瘤效应。
2.3PKGI与卵巢癌的关系
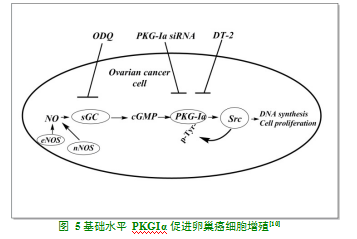
体外研究显示,在OV2008、A2780s和A2780卵巢癌细胞中,NO/sGC/cGMP/PKG信号通路的基础活性能够下调p53介导自发性凋亡,给予ODT(sGC抑制剂)能够延长p53半衰期,诱导p53丝氨酸15位磷酸化,上调p53依赖的基因产物,如p21、Bax等,诱导细胞凋亡[35]。进一步研究发现,给予ODT、DT-2(PKGIα特异性抑制剂)和基因沉默PKGIα能够抑制卵巢癌细胞的DNA合成和增殖,这提示cGMP/PKGIα的基础活性在促进卵巢癌细胞增殖方面有重要作用,可能机制如下:基础水平的NO(eNOS、nNOS催化产生)激活细胞内sGC,sGC水解GTP生成cGMP,基础水平的cGMP又可激活对cGMP敏感的PKGIα,PKGIα通过与Src相互作用,增强Src的激酶活性,进而引起DNA合成和细胞增殖(见图5)[10]。综上可知,PKG基础活性(尤其是PKGIα)可促进卵巢癌细胞DNA合成和增殖,抑制细胞的自发性凋亡,具有一定的促肿瘤效应。
2.4PKGI与其他肿瘤的关系
文献报道,PKGI信号通路在多种细胞中通过不同的机制,发挥细胞保护作用[36]。体外研究发现,在NCI-H460和A549非小细胞性肺癌细胞中,维持PKGIα的基础活性能够保持较高的CREB(cAMP response element binding)磷酸化水平,上调c-IAP1、livin、survivin和Mcl-1蛋白的表达,抑制细胞自发性凋亡和促进集落形成,给予DT-2或基因沉默PKGIα则可诱导细胞凋亡[8]。提示在非小细胞性肺癌中PKGIα具有一定的促肿瘤效应。研究显示,胰腺癌细胞(BXPC3、Capan-1、Dan-G、MiaPaCa、Panc1、ACBRI515、HPNPCC)中均表达功能性PKGI,给予DT-2选择性抑制PKGIα,能够引起胰腺癌细胞坏死,抑制细胞增殖和侵袭,这种抗肿瘤效应可能与下调ERK1/2和p38有关[11]。另有体内研究发现,在C57BL/6小鼠Panc02原位肿瘤模型中,DT3可通过抑制PKGI,减小原位肿瘤体积,抑制肿瘤的转移,此效应可能是通过下调GSK-3,P38和CREB磷酸化水平引起的[7]。综上,PKGI在非小细胞性肺癌和胰腺癌中也发挥着重要的生理作用且PKGIα显示促肿瘤效应。
3以cGMP/PKGI信号通路为靶点的抗肿瘤研究
研究发现,白芦藜醇可以抑制PKGIα的活性,下调Src磷酸化水平,抑制卵巢癌细胞DNA合成和细胞粘附[37]。白芦藜醇作为自然界广泛存在的天然产物,具有低毒多靶的特性,因此有望成为靶向PKGI的抗肿瘤药物。在结肠癌相关抗原分析中,发现PKGI与肿瘤的增殖和血管形成相关,通过合成肿瘤抗原PKGI的HLA-A2限制性表位肽,诱导HLA-A2限制性细胞毒性T淋巴细胞(CTL)的激活,激活的CTL可以靶向杀死体内的肿瘤细胞,发挥抗肿瘤效应[38]。因此,肿瘤PKGI表位肽有望成为靶向抗肿瘤多肽免疫治疗疫苗。近年研究证实,非甾体抗炎药(nonsteroidal anti-inflammatory drugs,NSAIDs)和环氧合酶-2(cyclooxygenase-2,COX-2)抑制剂具有潜在的抗肿瘤活性[39,40],但不幸的是,抑制环氧合酶(COX)可引起潜在的致命毒性反应。有研究显示,通过结构改造硫化舒林酸(一种非甾体抗炎药),得到化合物SSA,虽然SSA失去对COX-1和COX-2的抑制作用,但可以选择性抑制PDE5,增加乳腺癌细胞内cGMP的量,通过激活PKG(PKGIβ),下调Wnt/β-catenin,诱导细胞凋亡[41,42]。这提示化合物结构改造也是发现抗肿瘤药物的重要途径之一。另有体内研究报道,激活PKGI,可以通过降低Bax/Bcl2比值、caspase3活性和细胞色素c的释放,保护线粒体,缓解顺铂引起的肾损伤[42]。由此可见,PKGI在抗肿瘤的同时,还可以保护机体的重要器官,显示出广阔的药用前景。
4结语
近年来,随着研究的不断深入,PKGI信号通路与多种肿瘤的关系得到进一步阐明,为靶向PKGI信号通路肿瘤治疗提供了理论依据,但由于PKGI各亚型在不同肿瘤组织中的表达和活性的不同,目前针对PKGI信号通路的抗肿瘤药物仍处在研发阶段。通过文献综述可以对药物研发提供以下思路:(1)选择性,PKGI亚型的选择性抑制或激活(如选择性激活PKGIβ;选择性抑制PKGIα;选择性激活PKGIβ与选择性抑制PKGIα联用)对肿瘤治疗更有针对性;(2)药物骈合设计理念,大量研究显示通过联合用药,作用于PKGI通路的不同环节,可以很大程度增强药物的抗肿瘤活性(如顺铂和DT-2联用,D1R受体激动剂与PDE5的联用),因此可以通过药物骈合原理,将作用于PKGI通路不同环节的小分子化合物通过化学键骈合在一起,经体内酶代谢后释放出针对各靶点的活性化合物,发挥协同抗肿瘤作用;(3)天然产物,天然产物具有结构新颖,低毒等特点,是发现针对PKGI信号通路抗肿瘤的天然宝库;(4)药物结构改造,通过对已上市药物进行结构改造,减轻药物毒副作用,增强抗肿瘤活性,是发现潜在作用于PKGI信号通路抗肿瘤药物的有效途径之一。虽然PKGI与肿瘤之间的联系尚不十分明确,但可以肯定,针对PKGI信号通路的药物研发,将成为肿瘤治疗的有效方法。
参考文献
[1]Baker DA,Stewart LB,Large JM,et al.A potent series targeting the malarial cGMP-dependent protein kinase clears infection and blocks transmission[J].Nat Commun,2017,8(1):430.
[2]Hofmann F,Wegener JW.cGMP-dependent protein kinases(cGK)[J].Me-thods Mol Biol,2013,1020:17-50.
[3]Bryan NS,Bian K,Murad F.Discovery of the nitric oxide signaling pathway and targets for drug development[J].Front Biosci(Landmark Ed),2009,14,(1):1-18.
[4]El-Sehemy A,Chang AC,Azad AK,et al.Notch activation augments nitric oxide/soluble guanylyl cyclase signaling in immortalized ovarian surface epi-thelial cells and ovarian cancer cells[J].Cell Signal,2013,25,(12):2780-2787.
[5]Fallahian F,Karami-Tehrani F,Salami S.Induction of apoptosis by type Ibeta protein kinase G in the human breast cancer cell lines MCF-7 and MDA-MB-468[J].Cell Biochem Funct,2012,30,(3):183-190.
[6]Li N,Xi Y,Tinsley HN,et al.Sulindac selectively inhibits colon tumor cell growth by activating the cGMP/PKG pathway to suppress Wnt/beta-cate-nin signaling[J].Mol Cancer Ther,2013,12,(9):1848-1859.
[7]Soltek S,Karakhanova S,Golovastova M,et al.Anti-tumor properties of the cGMP/protein kinase G inhibitor DT3 in pancreatic adenocarcinoma[J].Naunyn Schmiedebergs Arch Pharmacol,2015,388,(11):1121-1128.
[8]Wong JC,Bathina M,Fiscus RR.Cyclic GMP/protein kinase G type-Ialpha(PKG-Ialpha)signaling pathway promotes CREB phosphorylation and maintains higher c-IAP1,livin,survivin,and Mcl-1 expression and the inhibi-tion of PKG-Ialpha kinase activity synergizes with cisplatin in non-small cell lung cancer cells[J].J Cell Biochem,2012,113,(11):3587-3598.
[9]Kwon IK,Wang R,Thangaraju M,et al.PKG inhibits TCF signaling in colon cancer cells by blocking beta-catenin expression and activating FOXO4[J].Oncogene,2010,29,(23):3423-3434.
[10]Leung EL,Wong JC,Johlfs MG,et al.Protein kinase G type Ialpha activity in human ovarian cancer cells significantly contributes to enhanced Src activation and DNA synthesis/cell proliferation[J].Mol Cancer Res,2010,8,(4):578-591.
[11]Karakhanova S,Golovastova M,Philippov PP,et al.Interlude of cGMP and cGMP/protein kinase G type 1 in pancreatic adenocarcinoma cells[J].Pancreas,2014,43,(5):784-794.
[12]Orstavik S,Natarajan V,Tasken K,et al.Characterization of the human gene encoding the type I alpha and type I beta cGMP-dependent protein kinase(PRKG1)[J].Genomics,1997,42,(2):311-318.
[13]3 Orstavik S,Solberg R,Tasken K,et al.Molecular cloning,cDNA structure,and chromosomal localization of the human type II cGMP-dependent protein kinase[J].Biochem Biophys Res Commun,1996,220,(3):759-765.
[14]Campbell JC,Henning P,Franz E,et al.Structural Basis of Analog Specificity in PKG I and II[J].ACS Chem Biol,2017,12,(9):2388-2398.
[15]Kots AY,Martin E,Sharina IG,et al.A short history of cGMP,guanylyl cyclases,and cGMP-dependent protein kinases[J].Handb Exp Pharmacol,2009,(191):1-14.
[16]Browning DD,Kwon IK,Wang R.cGMP-dependent protein kinases as po-tential targets for colon cancer prevention and treatment[J].Future Med Chem,2010,2,(1):65-80.
[17]Piwkowska A,Rogacka D,Kasztan M,et al.Insulin increases glomerular filtra-tion barrier permeability through dimerization of protein kinase G type Ial-pha subunits[J].Biochim Biophys Acta,2013,1832,(6):791-804.
[18]Joshi S,Nelson MT,Werner ME.Amplified NO/cGMP-mediated relaxation and ryanodine receptor-to-BKCa channel signalling in corpus cavernosum smooth muscle from phospholamban knockout mice[J].Br J Pharma-col,2012,165,(2):455-466.
[19]Wang XH,Tong M,Dou D,et al.[Role of cGMP-dependent protein kinase in the cardiovascular system][J].Sheng Li Ke Xue Jin Zhan,2005,36,(4):299-303.
[20]Wang Y,El-Zaru MR,Surks HK,et al.Formin homology domain protein(FHOD1)is a cyclic GMP-dependent protein kinase I-binding protein and substrate in vascular smooth muscle cells[J].J Biol Chem,2004,279,(23):24420-24426.
[21]Li R,Bourcy K,Wang T,et al.The involvement of vimentin in copper-induced regression of cardiomyocyte hypertrophy[J].Metallomics,2015,7(9):1331-1337.
[22]Lund N,Henrion D,Tiede P,et al.Vimentin expression influences flow de-pendent VASP phosphorylation and regulates cell migration and proliferation[J].Biochem Biophys Res Commun,2010,395,(3):401-406.
[23]Banday AA,Lokhandwala MF.Oxidative stress impairs cGMP-dependent protein kinase activation and vasodilator-stimulated phosphoprotein se-rine-phosphorylation[J].Clin Exp Hypertens,2019,41,(1):5-13.
[24]Nalli AD,Bhattacharya S,Wang H,et al.Augmentation of cGMP/PKG pathway and colonic motility by hydrogen sulfide[J].Am J Physiol Gastrointest Liver Physiol,2017,313,(4):330-341.
[25]Sellak H,Wu S,Lincoln TM.PKG induces the expression of tumor suppressor genes in colon cancer cells[J].Faseb Journal,2009,23(1):699-706.
[26]Kwon IK,Schoenlein PV,Delk J,et al.Expression of cyclic guanosine monophosphate-dependent protein kinase in metastatic colon carcinoma cells blocks tumor angiogenesis[J].Cancer,2008,112,(7):1462-1470.
[27]Hou Y,Wong E,Martin J,et al.A role for cyclic-GMP dependent protein kinase in anoikis[J].Cell Signal,2006,18,(6):882-888.
[28]Babykutty S,Suboj P,Srinivas P,et al.Insidious role of nitric oxide in migration/invasion of colon cancer cells by upregulating MMP-2/9 via activation of cGMP-PKG-ERK signaling pathways[J].Clin Exp Metastasis,2012,29,(5):471-492.
[29]Fiscus RR,Johlfs MG,Bathina M,et al.Mesothelioma and lung cancer cell lines differentially express the protein kinase G splice variants PKG-I alpha and PKG-I beta:Involvement in regulating apoptosis[J].Mol Cancer Ther 2007,6(12):3586-3587.
[30]Ho KK,Myatt SS,Lam EW.Many forks in the path:cycling with FoxO[J].Oncogene,2008,27,(16):2300-2311.
[31]Li N,Lee K,Xi Y,et al.Phosphodiesterase 10A:a novel target for selective inhibition of colon tumor cell growth and beta-catenin-dependent TCF transcriptional activity[J].Oncogene,2015,34,(12):1499-1509.
[32]Lee K,Lindsey AS,Li N,et al.beta-catenin nuclear translocation in colorectal cancer cells is suppressed by PDE10A inhibition,cGMP elevation,and activation of PKG[J].Oncotarget,2016,7,(5):5353-5365.
[33]Borcherding DC,Tong W,Hugo ER,et al.Expression and therapeutic targeting of dopamine receptor-1(D1R)in breast cancer[J].Oncogene,2016,35,(24):3103-3113.
[34]Schwappacher R,Rangaswami H,Su-Yuo J,et al.cGMP-dependent protein kinase Ibeta regulates breast cancer cell migration and invasion via interaction with the actin/myosin-associated protein caldesmon[J].J Cell Sci,2013,126,(7):1626-1636.
[35]Fraser M,Chan SL,Chan SS,et al.Regulation of p53 and suppression of apoptosis by the soluble guanylyl cyclase/cGMP pathway in human ovarian cancer cells[J].Oncogene,2006,25,(15):2203-2212.
[36]Francis SH,Busch JL,Corbin JD,et al.cGMP-dependent protein kinases and cGMP phosphodiesterases in nitric oxide and cGMP action[J].Pharmacol Rev,2010,62,(3):525-563.
[37]Gorjala P,Wong JC,Constantino BF,et al.Resveratrol alters the kinase activity of PKG-I alpha and Src family in A2780cp cells(ovarian cancer cell line with mutated p53)resulting in growth inhibition[J].Cancer Res 2014,74(19):4558.
[38]Shichijo S,Azuma K,Komatsu N,et al.Two proliferation-related proteins,TYMS and PGK1,could be new cytotoxic T lymphocyte-directed tumor-associated antigens of HLA-A2+colon cancer[J].Clin Cancer Res,2004,10,(17):5828-5836.
[39]Lee K,G AP.The interaction between the Wnt/beta-catenin signaling cascade and PKG activation in cancer[J].J Biomed Res,2017,31,(3):189-196.
[40]Doat S,Cenee S,Tretarre B,et al.Nonsteroidal anti-inflammatory drugs(NSAIDs)and prostate cancer risk:results from the EPICAP study[J].Cancer Med,2017,6,(10):2461-2470.
[41]Constance JE,Campbell SC,Somani AA,et al.Pharmacokinetics,pharmacodynamics and pharmacogenetics associated with nonsteroidal anti-inflammatory drugs and opioids in pediatric cancer patients[J].Expert Opin Drug Metab Toxicol,2017,13,(7):715-724.
[42]Derry S,Wiffen PJ,Moore RA,et al.Oral nonsteroidal anti-inflammatory drugs(NSAIDs)for cancer pain in adults[J].Cochrane Database Syst Rev,2017,7:Cd012638.
关注SCI论文创作发表,寻求SCI论文修改润色、SCI论文代发表等服务支撑,请锁定SCI论文网!
文章出自SCI论文网转载请注明出处:https://www.lunwensci.com/yixuelunwen/23331.html